|
In a few cases, the magnitude of the charge is equal to the group number, but in most cases it is not. The periodic table is less useful in predicting the most likely charge on cations formed by B-group elements. 
For a charge, however, we usually omit the 1 and write just Thus, and but rather than In writing chemical symbols of cations, we write the ionic charge as an Arabic numeral followed by a plus sign. To name monatomic cations, we add the word ion to the name of the parent element: sodium ion, magnesium ion, and so on. (3) The nonmetals of groups 7A and 6A, along with nitrogen and phosphorus of group 5A, form anions that have a charge equal to the group number minus 8. 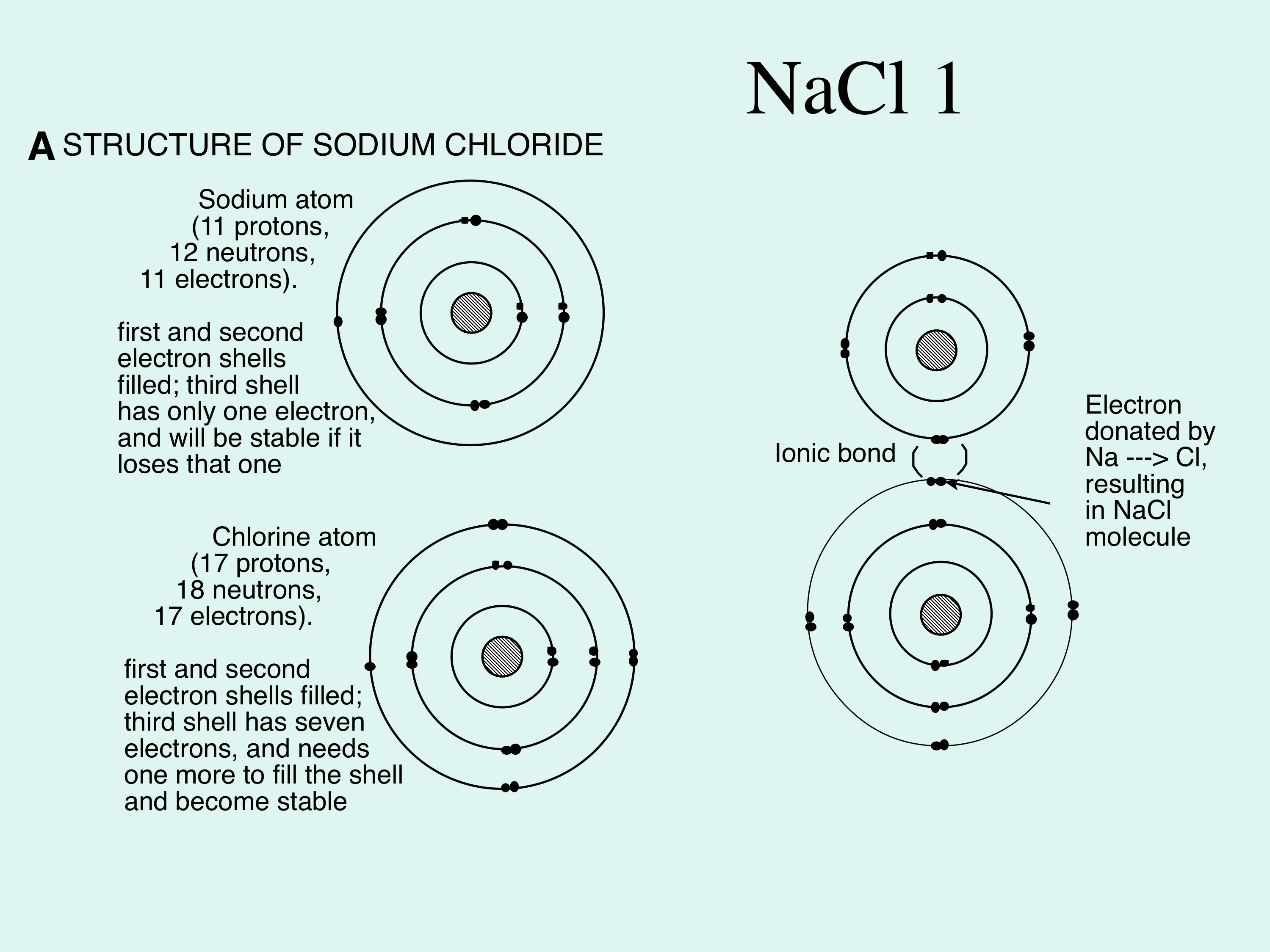
(2) Most of the metals of the B groups form two or more cations of different charges, though in some cases only one of these cations is commonly encountered. Three general observations can be made of these data: (1) Aluminum and the metals of groups 1A and 2A form just one cation, which carries a positive charge equal in magnitude to the A-group number. Aluminum, the most common and commercially important metal in group 3A, forms cations with charge.įIGURE 2.10 Symbols and periodic table locations of some monatomic ions Thus, the highly reactive metal atoms of group 1Athe alkali metalsgive up one electron to form cations with charge Group 2A atomsthe alkaline earth metalsgive up two electrons to form cations with charge. For metal atoms in the A groups, the number of electrons given up is usually equal to the periodic table group number. We can use the periodic table to predict the charges on some monatomic ions (Figure 2.10). In ionic compounds, there are no identifiable smallest entities comparable to the molecules of a molecular compound. Such clusters are called ionic compounds. Oppositely charged ions (cations and anions) attract one another and are held together in huge clusters by electrostatic attractions. 
For example, when the group of bonded atoms consisting of one sulfur atom and four oxygen atoms gains two electrons, the resulting ion is Some groupings of bonded atoms can also lose or gain electrons to form polyatomic ions. In general, metal atoms form cations and nonmetal atoms form anions. If a sodium atom loses one electron, it acquires a charge of and is represented as If a chlorine atom gains one electron, it acquires a charge of and is written as Positively charged ions are called cations, and negatively charged ions are called anions. Monatomic ions (also called simple ions) are formed when a single atom loses or gains one or more electrons. They can occur only in the presence of negatively charged ions. Sodium ions are generally unreactive, and they can't exist alone. Sodium atoms make up an element that, although quite reactive, can exist individually. Ions differ greatly from the atoms from which they are formed. If electrons are gained, there are more electrons than protons in the resulting ion and so it has a negative charge. If electrons are lost, there are more protons than electrons in the resulting ion and so it has a positive charge. Ions are formed only through the loss or gain of electrons there is no change in the number of protons in the nucleus of the atom(s). In some chemical reactions, however, an individual atom or a group of bonded atoms may lose or gain one or more electrons, thereby acquiring a net electric charge and becoming an ion. 
These names are based on their Greek words.In an isolated atom, the number of protons equals the number of electrons, and the atom is therefore electrically neutral. You have to remember which type of charged particle known by what name. Note: This question is totally memory based. Positron is a very stable particle where positron is a very unstable particle.įrom the above points we can conclude that the correct answer is “A”, all others are incorrect. And protons and positrons are two different things. Positron is a subatomic particle with the same mass as an electron and a numerically equal but positive charge. The ions of all non metal elements are anions. An anion contains more electrons than a normal atom. It is charged because the number of electrons do not equal to the number of protons in the atom or molecule. And an ion is a charged atom or molecule. Hint: From the given question we can take hints that the question is talking about an ion which has some charge on it.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
